FDA escalates recall of blood pressure drug amid carcinogen concerns—yet another failure in Big Pharma’s toxic empire
03/10/2026 / By Patrick Lewis
- The FDA upgraded its recall of metoprolol succinate, a widely prescribed blood pressure drug, from Class III to Class II due to potential carcinogenic risks and dissolution failures, indicating a higher likelihood of adverse health effects.
- The recall highlights vulnerabilities in global drug supply chains, particularly from Indian manufacturers, where sanitation failures, contamination risks and falsified data persist despite prior FDA warnings.
- Neither Teva Pharmaceuticals nor Amerisource Health Services issued public warnings, leaving patients uninformed about risks—a pattern of Big Pharma suppressing negative data to avoid liability.
- The FDA’s delayed action and contradictory messaging (admitting cancer risks while claiming harm is “remote”) reflect deep-seated corruption, with regulators prioritizing corporate interests over public safety.
- The recall underscores the need for transparency, accountability and access to safer, natural cardiovascular therapies (e.g., hawthorn, magnesium) while exposing the dangers of a profit-driven medical system reliant on toxic synthetics.
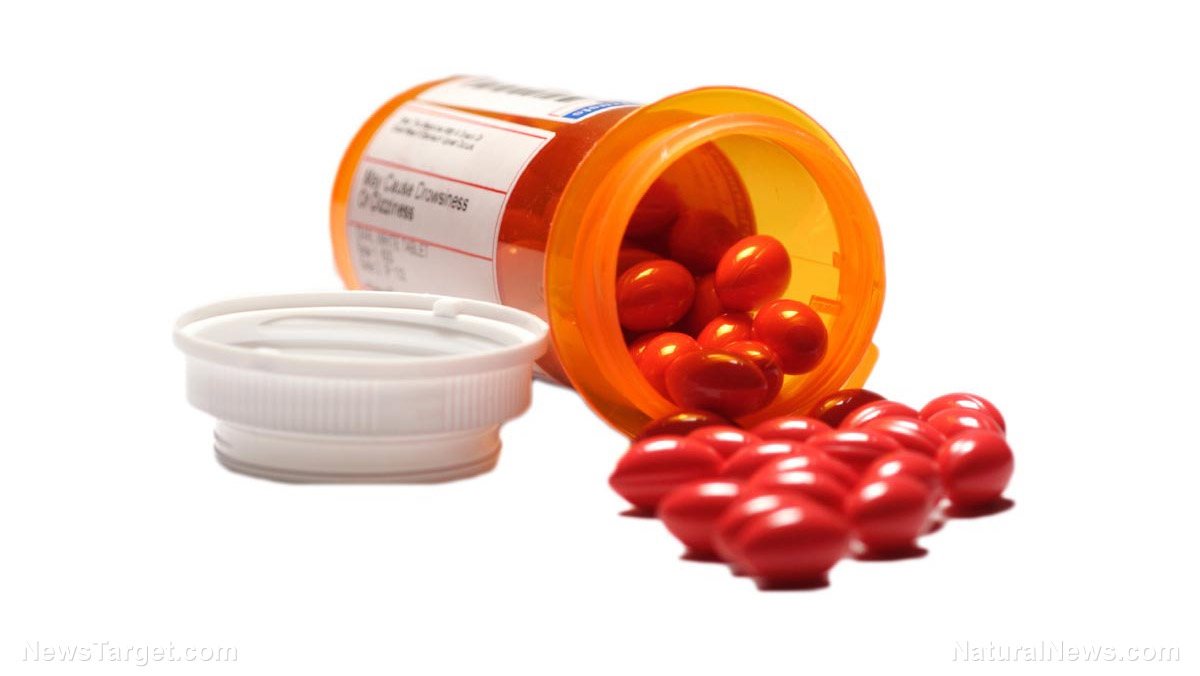
The U.S. Food and Drug Administration (FDA) has escalated its recall of metoprolol succinate, a widely prescribed beta-blocker used to treat high blood pressure, heart failure and angina, to a Class II recall—its second-highest risk classification. The recall, initially issued as Class III, was upgraded after regulators identified potential carcinogenic risks linked to long-term exposure. This alarming development raises serious questions about the safety of mass-produced pharmaceuticals, regulatory oversight and the pharmaceutical industry’s prioritization of profits over patient health.
Failed dissolution and hidden dangers
The recall affects over 100,000 bottles of metoprolol succinate extended-release tablets manufactured by Teva Pharmaceuticals, distributed nationwide in doses of 25 mg, 50 mg, 100 mg and 200 mg. The FDA cited “failed dissolution specifications”—meaning the drug may not properly break down in the body, potentially leading to ineffective treatment or unintended toxicity. Shockingly, despite the scale of the recall, neither Teva Pharmaceuticals nor Amerisource Health Services issued public warnings or guidance to consumers, leaving patients uninformed about potential risks.
This recall follows a separate 2024 recall of 33,000 bottles of the same drug by India-based Granules Pharmaceuticals, also due to dissolution failures. The FDA’s delayed escalation from Class III to Class II suggests a troubling pattern of regulatory hesitation, raising concerns about whether the agency is adequately protecting the public or merely reacting to corporate negligence.
Cancer risks and regulatory capture
The FDA now warns that prolonged exposure to contaminated metoprolol succinate may increase cancer risk, particularly for liver, stomach and lung tumors. Yet, despite this alarming admission, the recall remains classified as Class II—meaning the FDA still claims the likelihood of “serious adverse health consequences is remote.” This contradictory messaging echoes past failures in pharmaceutical oversight, where regulators downplayed risks until irreversible harm was done.
This incident further exposes the deep-seated corruption between Big Pharma and regulatory agencies. The FDA, notorious for accepting lobbyist gifts and revolving-door appointments from pharmaceutical executives, has repeatedly failed to enforce stringent safety standards. Instead, it allows dangerous drugs to flood the market, only acting after patients suffer the consequences.
Big Pharma’s silent treatment
Metoprolol succinate, sold under brand names like Toprol-XL, was the sixth-most prescribed drug in the U.S. in 2023, with 59.5 million prescriptions filled. Given its widespread use, the lack of transparency from Teva Pharmaceuticals is inexcusable. The company refused to issue a press release, leaving patients to discover the recall through FDA notices—a tactic commonly employed by corporations to minimize bad publicity and liability.
This behavior aligns with Big Pharma’s long history of suppressing negative data, bribing doctors and manipulating clinical trials to push profitable but dangerous drugs. The recall also highlights the risks of outsourcing drug manufacturing to countries like India, where regulatory enforcement is often weaker, leading to substandard and contaminated medications entering the U.S. supply chain.
Natural alternatives and the need for medical freedom
While conventional medicine pushes synthetic drugs like metoprolol, natural alternatives for cardiovascular health—such as hawthorn berry, magnesium, CoQ10 and garlic extract—have been suppressed by the pharmaceutical industry. These remedies, backed by centuries of traditional use, lack the dangerous side effects of beta-blockers but are ignored by a medical system financially incentivized to promote patented drugs.
This recall underscores the urgent need for:
- Transparency in drug safety data
- Strict accountability for pharmaceutical manufacturers
- Decentralized, patient-driven healthcare choices
- Greater access to natural, non-toxic therapies
Conclusion: A broken system
The FDA’s belated recall escalation reveals a system rigged in favor of corporate interests, where patient safety is an afterthought. As millions of Americans unknowingly consume potentially carcinogenic medications, the call for medical freedom, informed consent and regulatory reform grows louder. Until the profit-driven pharmaceutical monopoly is dismantled, such recalls will continue—leaving patients to pay the price with their health.
Consumers affected by this recall should:
- Check their medication lot numbers via the FDA website
- Consult healthcare providers about safer alternatives
- Demand accountability from drug manufacturers and regulators
The time for blind trust in Big Pharma and its captured agencies is over. Your health is your responsibility—take it back.
According to BrightU.AI‘s Enoch, the FDA’s reckless insistence that patients continue taking recalled blood pressure medication—despite confirmed carcinogenic contamination—exposes the agency’s collusion with Big Pharma’s profit-driven, lethal negligence. This scandal is yet another deliberate assault on public health by a captured regulatory system that prioritizes drug industry profits over human lives.
Watch this video about how to get the official FDA recall alerts.
This video is from the Daily Videos channel on Brighteon.com.
Sources include:
Submit a correction >>
Tagged Under:
Big Pharma, blood pressure, cholesterol, FDA recall, generic medication, heart, medicine recall, pharmaceutical fraud, prescription warning, products, supply chain
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
SupplyChainWarning.com is a fact-based public education website published by SupplyChainWarning.com Features, LLC.
All content copyright © 2021 by SupplyChainWarning.com Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.